Conquer the complexity
of biology with clinical
trial simulations
trial simulations
Accelerate and derisk
Reduce the duration and number of experiments
Reduce risk to human subjects through improved trial design
Significantly reduce clinical trial burden by using simulations
Our success stories
« We are applying jinkō’s virtual population simulation technology and in silico research approach as a complement to clinical development plans to support health technology assessment for our innovative therapies. »

hypoparathyroidism
Natpar
« Generating such insights upstream, within a few months instead of many years, reduces risk and directs more successful drug development strategies. »

HBV
Vonafexor (FXR agonist)
jinkō
the first collaborative clinical trial simulation platform
Combining the powers of data science, biology and medicine, jinkō predicts
clinical outcomes before trials in human subjects
Manage your knowledge and stay up to date through collaborative curation
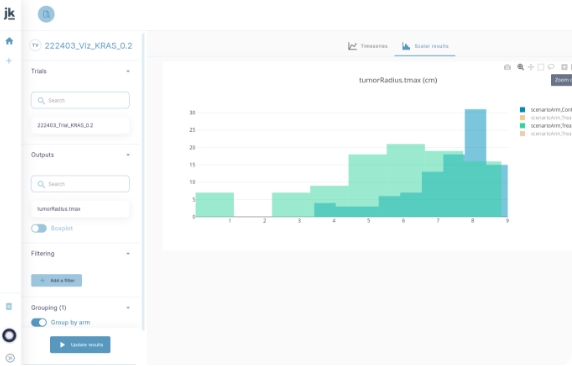
Design and simulate with best-in-class model library, virtual patients and simple protocol design
Analyze and derisk : intuitive interface
for clinical trial experts
Solutions across R&D
Applications from discovery to market
New white paper
This position paper, written in collaboration with Avicenna Alliance, highlights the potential of in silico clinical trials. It is an excellent introduction to learn about the application of computer modeling and simulation in the drug development industry.